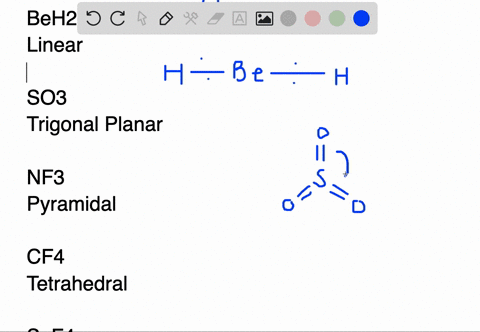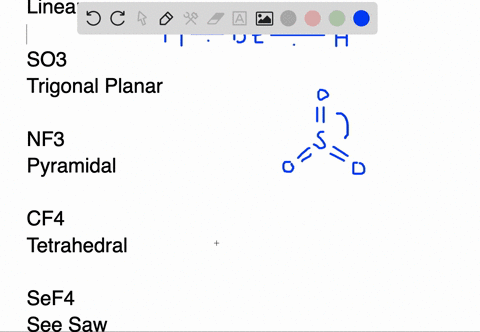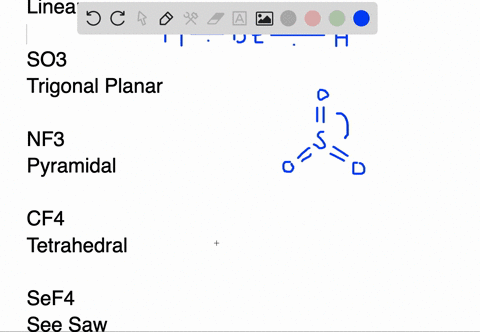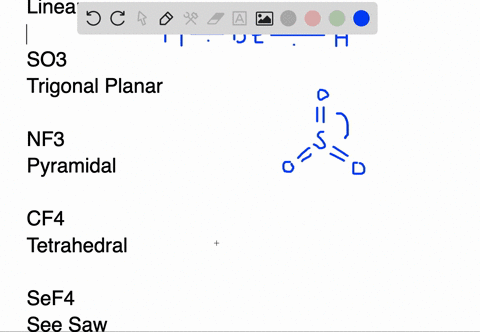
BeH2 Lewis Structure (Beryllium Hydride) | BeH2 Lewis Structure (Beryllium Hydride) Beryllium Hydride or BeH2 has a simple arrangement of atoms. In this video, we help you find out the Lewis dot...

SOLVED: 2A) Which of the folloiwng molecules would be considered non-polar? BeH2, Br2S, PF3, TeCl2, HCL – show all work
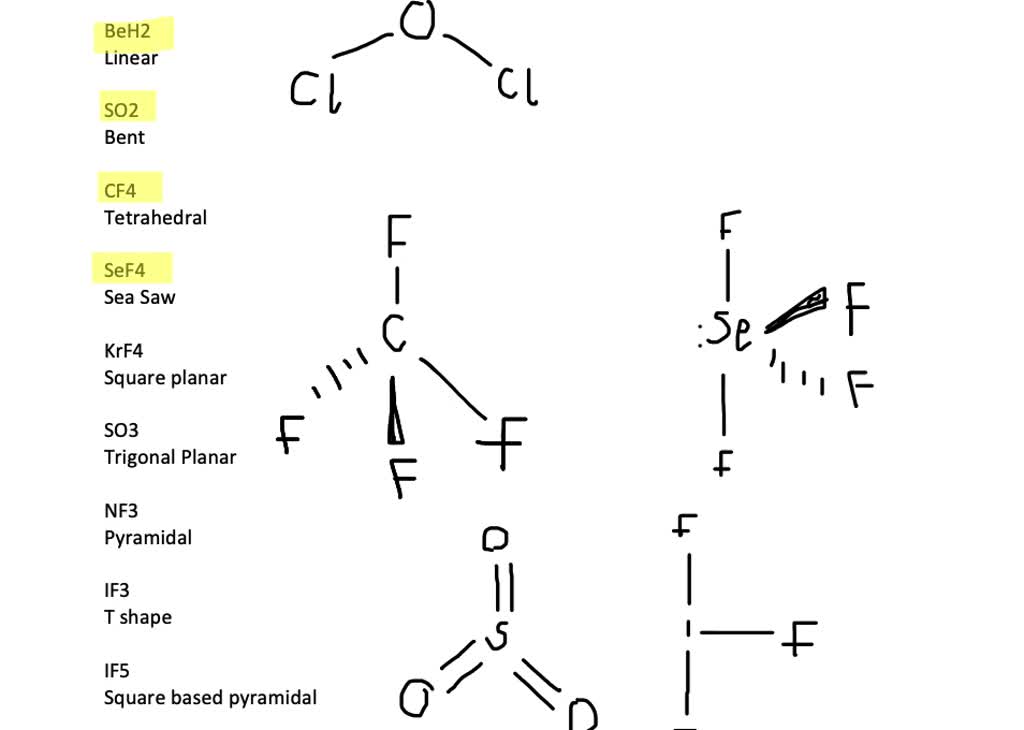
SOLVED:Draw Lewis structures and predict the molecular structures of the following. (See Exercises 89 and 90 . ) a. OCl2, KrF2, BeH2, SO2 c. CF4, SeF4, KrF4 b. SO3, NF3, IF3 d.

Review Polar Covalent: unequal sharing of electrons (electronegativity difference >0.4) Nonpolar Covalent: equal sharing of electrons (electronegativity. - ppt download

SOLVED:The bonds in beryllium hydride (BeH 2 ) molecules are polar, and yet the dipole moment of the molecule is zero. Explain.

How many of the following compounds are non- polar? NH3 ,NF3 ,BeH2 ,CO2 ,H2O ,HI, H2 ,BF3 ,CH4 CHCl3 ,CH2Cl2 , CCl4