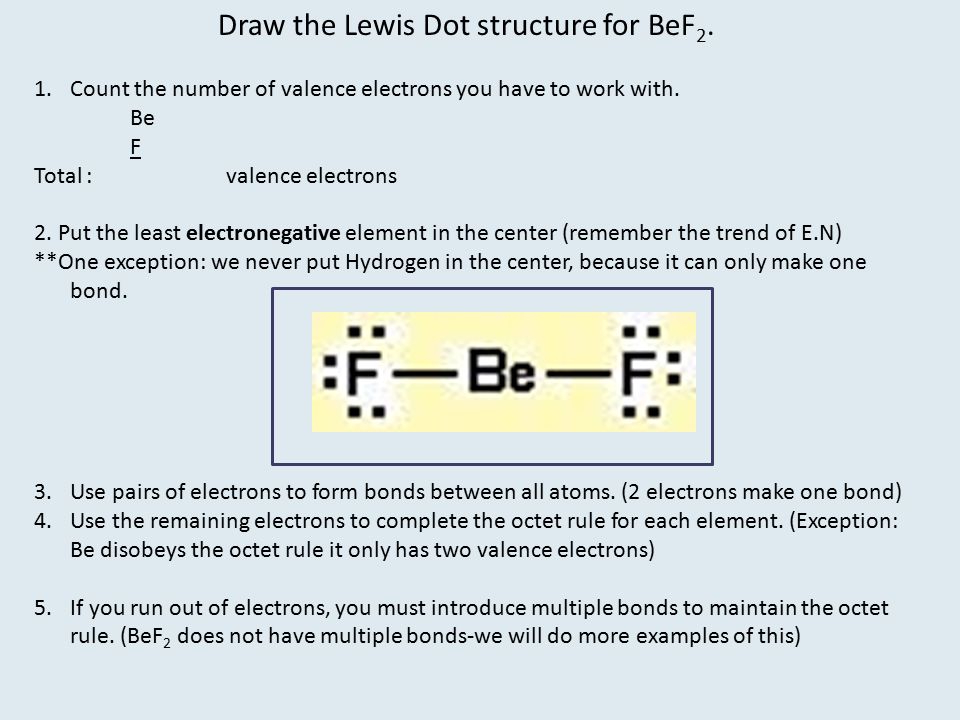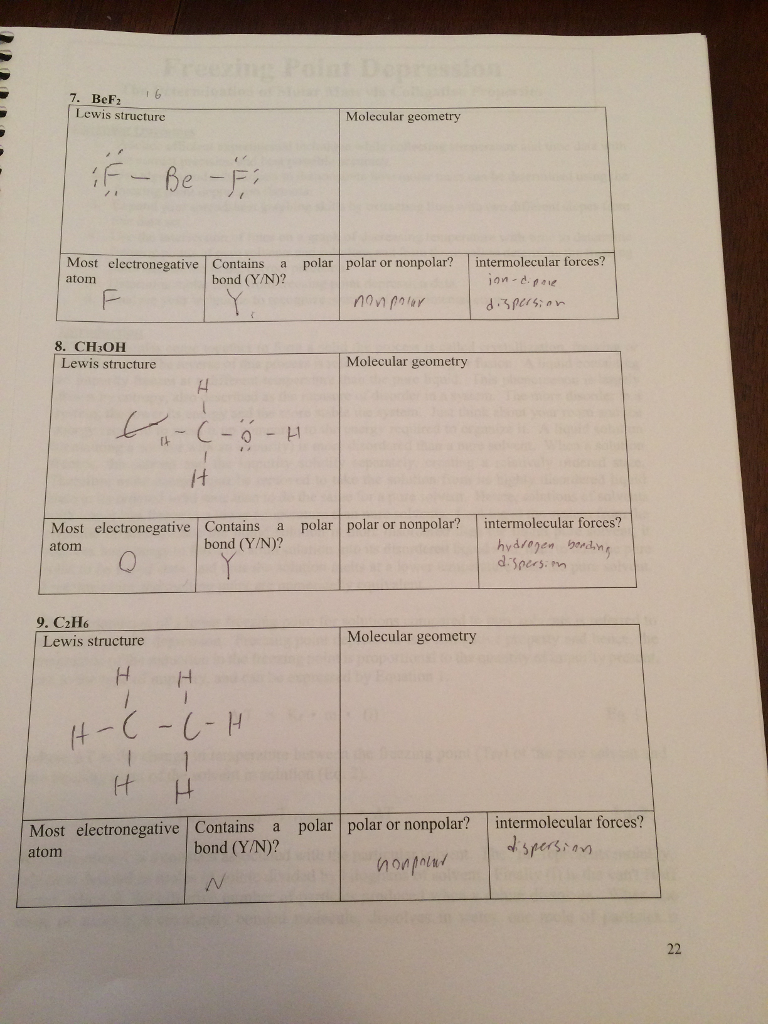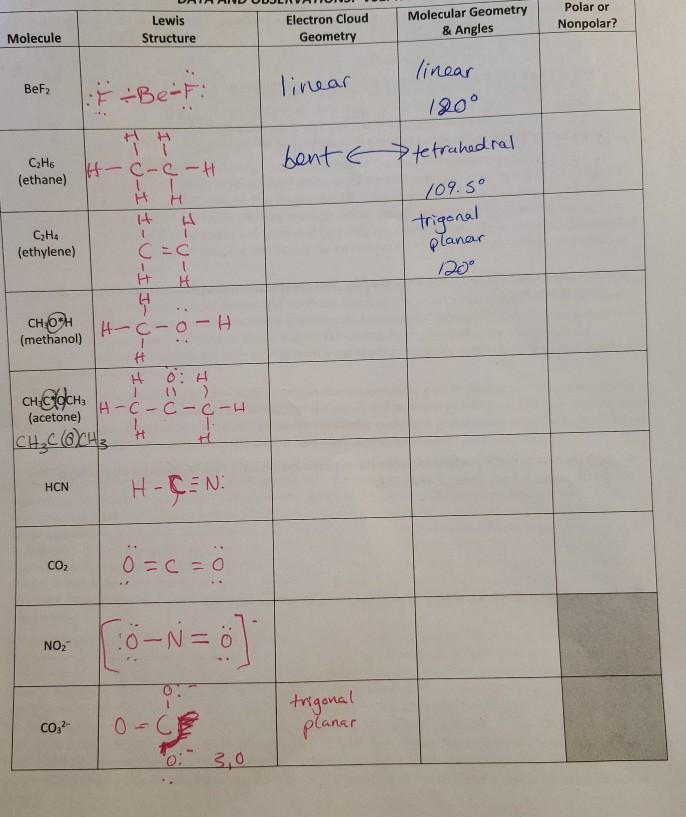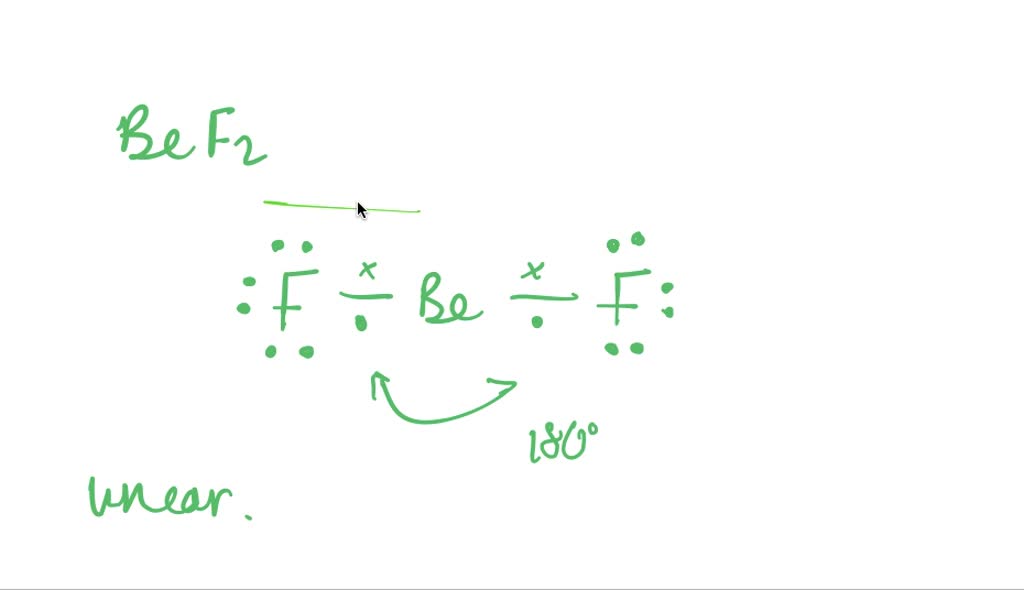
SOLVED:Despite the large electronegativity difference between fluorine and beryllium atoms, BeF2 forms linear molecules in gas phase. Are these molecules polar or nonpolar? Explain.

PS 15 2020 - Chem 105 - Problem Set 15 – Molecular Shape, Polarity Chem 105 Once you know the - StuDocu